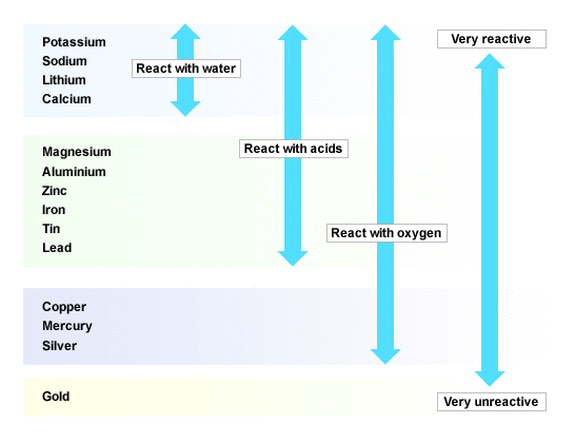
Compare the reactivity of the two metals.Into the other, drop a small piece of calcium.Into one test-tube drop a small piece of magnesium.Fill two test-tubes a quarter with dilute hydrochloric acid.Calcium is water reactive, see CLEAPSS Hazcard HC019c.Magnesium is pyrophoric and water reactive, see CLEAPSS Hazcard HC059b.Hydrochloric acid is of low hazard, see CLEAPSS Hazcard HC047a.Read our standard health and safety guidance.Students will be able to discover that group 1 metals are more reactive than group 2 RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation.The activity series determines the capability to displace hydrogen gas from water and other solutions like acid or ionic solutions. Lithium is a highly active element that reacts with steam and acids. Another example is the reaction of lithium with cold water as it displaces hydrogen from the water. It shows that it does not react with almost anything, so it is safe to use as ornaments or jewelry. One of the biggest examples of activity series is the noble metals like silver, gold, and platinum, which are found at the bottom of the metal activity series. The activity series is used to determine how an element will react when it is kept in water, acid, or solutions containing different ions of different metals.Īn Activity series consists of lists of elements according to their decreasing order of reactivity. As we know that the highly reactive elements will replace the less reactive elements hence, you will get the products of the chemical reaction. 
Also, check the reaction of the elements if it is highly active or not. See where the elements are situated in the Activity Series. Step 1: Check the elements in the reaction. We need to understand the Activity Series to determine and predict the products of the reaction. 
Some of the elements do not react at all these are the metals that are under hydrogen, like silver, gold, and platinum. Also, to visualize which metals take time to react to replace the hydrogen atoms like cobalt, nickel, tin, etc. The activity series of metals was developed so that it becomes easier for us to understand which metals are more reactive and which ones will react in any medium like sodium, magnesium, etc. The faster the reaction, the faster it loses electrons to form positive charges or positive ions. In general, the more reactive a metal is, the more vigorously it reacts with other metals elements. The reactivity series of metals is a chart listing metals in order of decreasing reactivity of the elements. The activity series is a set of different elements like metals and non-metals listed in decreasing order of their reactivity. It helps to determine the nature of the reaction and which element will displace the other element in a chemical reaction. The Activity Series is developed to determine the reaction of metals and non-metals elements with different elements in different conditions. Well, it can be difficult to understand how an element will react and in which conditions hence, to know the reactivity of metals, the activity series is developed. But, how to understand which elements will react with the other or which one will replace and take its place? We know due to some of the chemical reactions, all the elements react to form new compounds. Have you ever wondered why different metals and nonmetal elements react with each other? Why do some of the elements in the periodic table not react at all? All of these are very thoughtful questions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
